Real-World Data Collection and Patient Engagement
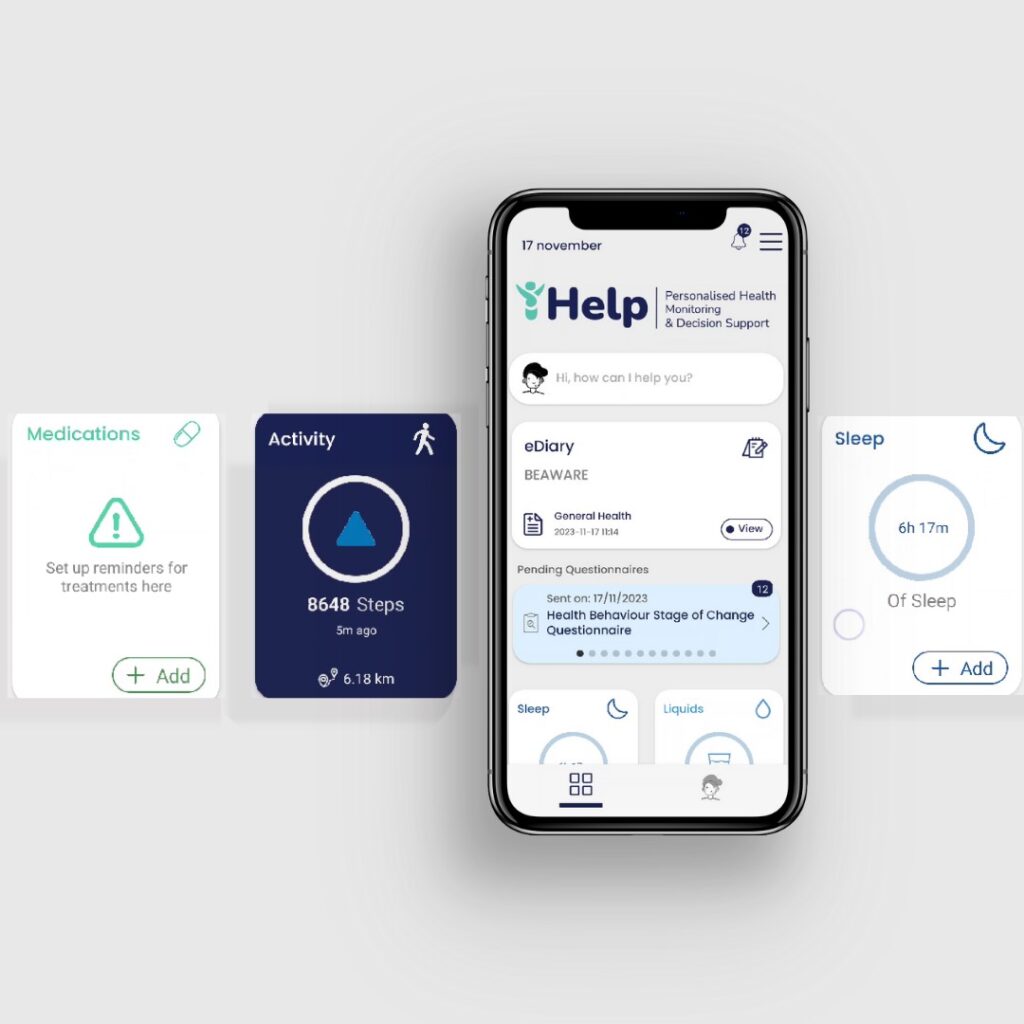
All iHelp patients experience the mobile application of Healthentia as their gateway in iHelp. Healhtentia is a medical device implemented as a digital platform for decentralized clinical trials, a product of the iHelp partner Innovation Sprint, a Belgian-based SME that brings innovative technologies in Life Sciences & MedTech to improve clinical trials and patients’ health. The mobile app acts as a patient companion, offering the necessary advice in the form of dialogues, and facilitating secondary data capture, a fundamental step for all five different pilots.
Patients engage with the Healthentia mobile app to seamlessly collect RWD from wearable devices tracking physiological and activity data. The app also incorporates subjective self-assessment through questionnaires. Simultaneously, healthcare professionals use this data to manage studies, define desired data attributes, and examine patients’ RWD. The iHelp Project spans various pilots, each addressing different aspects of pancreatic cancer—prevention, early detection, and disease progression monitoring. Since clinical partners have distinct data requirements, Healthentia adapts RWD collection and processing while employing different questionnaires tailored to each use case.
Measurements and Questionnaires
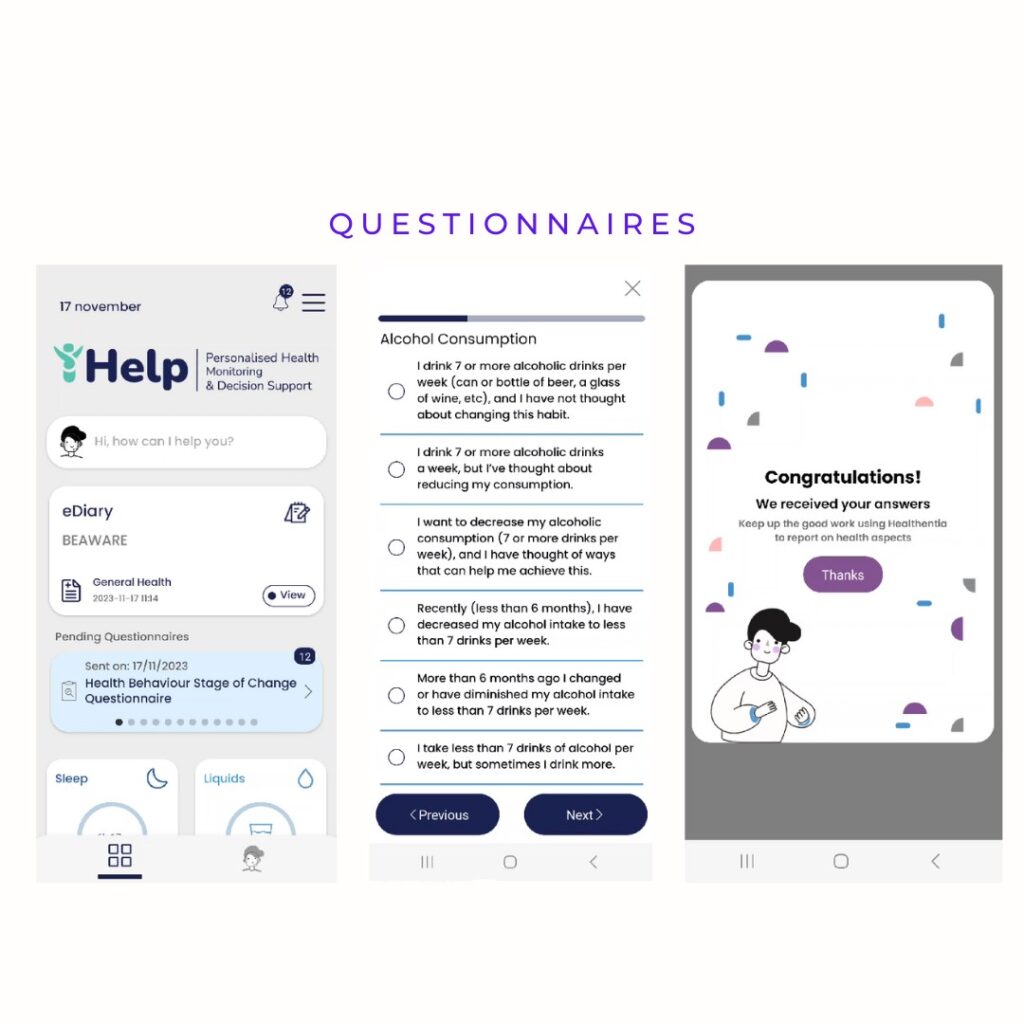
The standardized automated measurements cover physical activity, exercise sessions, heart rate info, and sleep info. Activity trackers (Garmin, Fitbit) and mobile phones (Android and iPhone sensors) seamlessly gather this data. Healthentia introduces 11 widgets, allowing study administrators to enable specific functionalities. For iHelp, four essential widgets—sleep, liquids, food categories, and e-diary—are integrated to enhance the depth of data insights. The extensive iHelp questionnaires, addressing aspects like sleep quality, mental well-being, and cancer symptoms, are scheduled at regular intervals and can be conveniently answered using the Healthentia app.
Virtual Coaching
Healthentia serves as a virtual coaching companion for patients, fostering a continuous dialogue aimed at enhancing their well-being. Daily or weekly notifications, strategically aligned with their doctor’s recommendations, inform patients about their progress and goals toward a healthier life. The virtual coach not only guides users on navigating the app and checking their set goals but also imparts valuable insights into the significance of various lifestyle aspects—such as nutrition, sleep quantity and quality, and exercise—in managing their disease.

The coach provides practical tips to facilitate lifestyle adjustments, making daily life and coping more manageable for the user. Importantly, patients have the option to initiate conversations with the virtual coach, creating a personalized and dynamic interaction. Every exchange between the virtual coach and the patient is meticulously recorded for future reference.
This recorded interaction serves as an invaluable source of secondary data for clinicians. By delving into these exchanges, clinicians gain deeper insights into their patients’ experiences, enabling them to tailor notifications, adjust goals on specific topics, and effectively manage their cohort of individuals. In essence, the virtual coaching component of Healthentia not only empowers patients in their health journey but also provides clinicians with a nuanced understanding of patient needs, facilitating more personalized and impactful healthcare interventions.
As we witness the ongoing evolution of data capture, analysis and action, the collaborative efforts of the iHelp partners open the way for a deeper understanding of pancreatic cancer and, ultimately, more effective preventive and therapeutic interventions. Stay tuned for more insights as we continue our journey through the iHelp Project.






